सभी उत्पाद देखने के लिए क्लिक करें Our Company
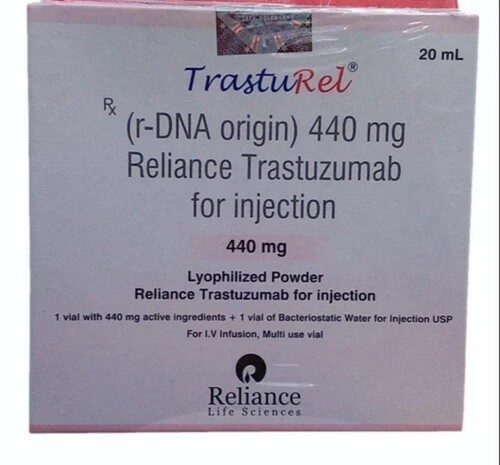
Price 10000 आईएनआर/ Vial
MOQ : 2-3 Vials
Specification
- खुराक
- 440 mg/vial
- परख
- As per label claim
- कैस नं
- 180288-69-1
- ग्रेड
- Pharmaceutical Grade
- समय सीमा समाप्ति की तारीख
- As per pack
- खुराक प्रपत्र
- Injection
- दवा का प्रकार
- Allopathic
- स्टोरेज
- Store at 2°C - 8°C (Refrigerated, Do Not Freeze)
- उपयोग
- For intravenous infusion only
- संकेत
- Treatment of HER2-positive breast cancer, metastatic gastric cancer
- मेडिसिन की उत्पत्ति
- India
- पैकेजिंग (मात्रा प्रति बॉक्स)
- 1 Vial + 1 Solvent
- नमक की संरचना
- Trastuzumab 440 mg
- आण्विक सूत्र
- C6470H9956N1712O2013S42
- दिखावट
- Lyophilized powder for injection
- Route of Administration
- Intravenous
- Brand Name
- TRASTUREL
- Preservation
- Contains Bacteriostatic Water
- Packaging Type
- Injection vial with solvent
- Therapeutic Class
- Antineoplastic (Monoclonal Antibody)
- Reconstitution Solvent
- 20 ml Bacteriostatic Water for Injection (BWFI)
- Precautions
- Administer under supervision of qualified healthcare professional
- Prescription/Non Prescription
- Prescription
Tell us about your requirement

Price: Â
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
मोबाइल number
Email
अधिक Products in एचआईवी एड्स दवा Category
25 मिलीग्राम टेनोफोवीर अलाफेनमाइड टैबलेट आईपी
मूल्य की इकाई : पैक/पैक
उपयोग : कंपनी के मानदंडों के अनुसार
मूल्य या मूल्य सीमा : आईएनआर
न्यूनतम आदेश मात्रा : 10
स्टोरेज : सूखी जगह
माप की इकाई : पैक/पैक


 जांच भेजें
जांच भेजें



 मुझे निःशुल्क कॉल करें
मुझे निःशुल्क कॉल करें
